A lot of confusion surrounds the use of the word Quantum. People have latched onto it to mean all sorts of weird things, which physicists quite rightly complain about – however if you ask them to explain it they invariably throw up equations, which are meaningless to almost anyone who doesn’t study the subject.
This is because phyicists use equations to explain what happens, but equations don’t explain why it happens. Nobody understands why it happens – or how it can.
To understand what happens in a nutshell it’s probaby best to begin with an electron, which is the small particle responsible for a flow of electricity, and the creation of the electromagnetic fields that power most of the things around us these days.
Electrons form part of atoms, and the configuration of electrons in an atom has a profound effect on the chemical properties of a substance. As part of an atom, electrons are attracted to the positive charge of protons in the nucleus. ‘Like’ charges (+/+ and -/-) repel, ‘unlike’ charges (+/-) attract. This is also true of magnets, where pairs of North poles and pairs of South poles repel but North and South poles attract, which is how a magnetic compass works because the Earth has a magnetic field.
The difference between electric charges and magnetic poles is that magnetic poles always occur in matched pairs (dipoles). There are two ends of a magnet – you never get just a North pole. You can however get separate ‘loose’ electrical charges, in the form of electrons and protons. Nobody knows why, especially as the electric and magnetic fields are so closely coupled, and energy travels in the form of electromagnetic waves which contain both electrical and magnetic components.
Physicists often treat the electron as a point particle because it is so small that the actual size of it is irrelevant in most calculations. It is also one of the fundamental particles, meaning that it isn’t made of anything else. Many other atomic particles seem to be formed from yet smaller ones, but the electron has been shown to be so perfectly featureless and symmetrical that it can’t conceivably have a complex internal structure[1]. This is important because it means that nothing can be ‘hiding’ in it.
Given that the two components are so closely coupled, it might therefore come as no surprise to discover that the electron is affected by both electric ‘fields’ (in other words charge) and also by magnetic fields. Its behaviour however can be very strange and unintuitive when you consider an electron in isolation – that is away from anything else.
For our purposes that’s exactly where we want to start, with a ‘free’ electron, somewhere out in space where it isn’t affected (very much) by anything else.
Our electron has properties which describe it. It has a charge, what we have called a negative charge, and it has mass and therefore energy, but these are the same for every electron. In this respect, every electron is exactly the same as every other electron. There is no way to tell them apart.
These values, or quantities which describe something such as an electron, are its Quantum Numbers. Quanta meaning amount. Generally, the quantum number is defined as being the parameter, and its value is the quantum state. So far we’ve seen two, and they are have fixed values.
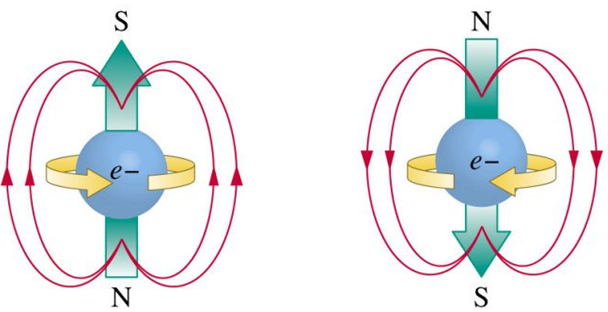
I’m now going to tell you something which is not true. Just like the Earth, electrons spin and they have magnetic poles. This is not surprising because electric and magnetic fields are inextricably connected. If spins, it has to spin about an axis, and any magnetic effect has to have two opposite poles.
The problem is that electrons don’t spin. They behave as if they do spin, in the sense they have angular momentum, like a spinning top or anything else that rotates, but they don’t really spin. There’s nothing to spin, they’re just a dot of something (Fig 2) which is actually fuzzy.

Okay, you may say, so it has a charge and a mass (which is energy), and also an intrinsic magnetic dipole moment. So there are three quantum numbers. It’s directional, so if you put an electron in a magnetic field it will line up with it, like a compass does in the Earth’s magnetic field. However, the problem with the magnetic dipole is interesting, and we’re finally getting closer to the big revelation which turned early 20th century physics on its head.
Unlike a magnetic compass, it can align with the external field in either direction – so it can line up North to North (parallel with the applied field lines) or the other way up.
So now we have three parameters, or quantum numbers, namely charge, energy, and magnetic direction. The killer in that previous sentence is the appearance of the concept of direction. Charge and energy have no direction. Suddenly we have a magnetic property, which has a direction because there has to be two poles.
Any given electron will either align itself up or down in an external magnetic field. This is repeatable. It has a memory of which direction its dipole will interact with a magnetic field – and that remains even when the external field is taken away and subsequently reapplied.
For various reasons, this ‘magnetic direction’ was associated with the direction of spin. We know spin is the same ‘quantity’ for every electron – because the calculation of their energy involves the apparent spin speed. Whether the electron points upwards or downwards in a magnetic field was attributed to the direction of spin. And here we have that word again. Direction. If it spins one way, it will align with an applied magnetic field North-to-North. If it spins the other way it will align North-to-South. The problem with that description is defining what is meant by one way and the other way. If you look at the Earth from the top it turns anti-clockwise. If you look at it from the bottom it turns clockwise. Now we’re saying top and bottom, as if there’s come universal definition of which way is up. There isn’t. Speaking of direction implies relative to something else.
There is no way to tell which way the electron in Fig 2 is ‘spinning’. It is featureless, so even if it were an animation of an electron it wouldn’t look any different. You can only define up and down, or top and bottom, by taking advantage of looking at its magnetic properties, and when you do that you’re introducing something which you can use as a relative direction.
So we now have three characteristics – quantum numbers, and their values – that appear to define an electron: Its charge and energy, which are constant – the same for every electron – and its spin. Well, the amount of spin is the same for every electron, and it forms part of the definition of it’s energy – you can’t change that – so the value of spin is also a constant. The variable thing is the direction of spin (Fig. 3). It’s variously quoted as plus or minus, or as spin-up or spin-down.
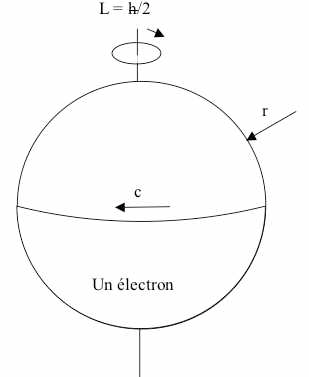
Considering the magnetic alignment as being related to the direction of spin makes sense, because it is something which you can imagine an electron physically having, like its charge and energy (mass). If we had a chance to reassign concepts to quantum numbers, it might make more sense to define the value of spin and the direction of spin as two separate quantities because one is variable and the other is not, but as they say: It is the way it is. The quantum values for spin are ±1/2.
So that would seem to settle it. There are only two kinds of electron. Electrons are only distinguishable from each other by the direction of their spin, and that can only be determined by placing them somewhere that allows you to define what you mean by a direction.
Side by side will do. That would allow you to determine that they either had the same spin direction or opposite spin directions but wouldn't tell you which one was up or down. This has implications in atomic structure because electrons in an atom like to be in pairs with opposite spins - it's a lower-energy state.
That’s what Quantum Numbers and Quantum Values are. Now let’s look at Entanglement.
Quantum Entanglement
We’ve spent a long time going on about the importance of direction, and how it affects our ability to define a quantum value such as spin. We also made a big deal about how spin makes electrons different.
There’s one more thing to understand before a concept of quantum entanglement becomes understandable. You could probably get away without it, but it won’t take long.
There is something special about the values of spin. Particles such as electrons with a non-integer spin (1/2) are called Fermions, and they cannot exist in the same place at the same time. Particles with an integer spin (0,1) are Bosons, and they can.
We defined electron spin as 1/2, so electrons are Fermions, and can’t exist in the same place at the same time – they’ll bump into each other.
However, if you get particles like electrons close enough together, by forcing them to be part of an atom for example, opposite spins will cancel out and they’ll behave as if they are one particle with a zero spin – which would be a Boson.
This is the ciritcal part to understanding about quantum entanglement.
Suppose we do something involving a particle collision which creates two electrons. They shoot off in different directions. The two electrons must have opposite spins – they were made in the same place and they wouldn’t have been able to exist there together if they both had the same spin.
If we look at those two electrons they do indeed have opposite spins. On the face of it this sounds fine. The thing which throws a spanner in the works is this: We discovered that both electrons could have either direction of spin, until you look at one of them, and then the spin direction of the other electron is determined. Not only does this happen instantly, it doesn’t matter how far apart they are. Once you find out what the spin of one of them is – the other instantly has the opposite spin.
Why? Nobody knows. Of course, we knew they had to have opposite spins but we just assumed this would be determined when they were created, not when you looked at one of them, which somehow ‘magically’ determined what the other one had to be. It is ‘spooky’ action at a distance and Einstein for one didn’t believe it. He thought they must be created with defined spins and we were missing something, somehow, in the methods we used to determine spin. After all, how do you know that the spin isn’t already defined before you look at it, and of course when you look at the other one it has the opposite spin.
“Dr. Bertlemann likes to wear two socks of different colours. Which colour he will have on a given foot on a given day is quite unpredictable. When you see that the first sock is pink you can be already sure that the second sock will not be pink. Observation of the first, and experience of Bertlemann, gives immediate information about the second.”[4]
This seems similar to our observation of quantum-entangled spin, however in the case of Dr. Bertlemann, the choice of which colour sock went on which foot was determined when he put his socks on. It is a predetermined outcome. This is not true of quantum entanglement. At their creation, neither electron has a direction of spin. In the place they were created, there is no concept of a universal direction. The spins are opposite directions relative to each other, but which one is spin-up and which one is spin-down is meaningless, because the concept of spin-up and spin-down rely on there being a magnetic reference to define a direction.
Just as there is no concept of speed at an instant in time (The Train and The Fly) there is no external concept of a spin direction in the place where the particles were created. All that can be said is that they are opposite to each other.
Entanglement was demonstrated to really occur in 1949, and the search was on for decades to devise some way to explain the correlation without having to admit that particles could exist in some form of half-defined form and synchronise their quantum states over a distance instantaneously[5].
Prior to this it was believed that the parameters (the quantum numbers) of particles were somehow kept locally – in other words in the particles. This realisation meant that the direction of spin is held within the particle – because it remembers it once defined – but the particle was not created with such a definition. The definition of spin direction was only created when it was first observed. In other words, when it was put in a situation where there was a concept of direction and the magnetic dipole needed to align in one direction or the other.
The mind-boggling realisation was that doing this to one of the pair instantly resulted in the other acquiring a spin direction parameter set to the opposite direction – almost as if touching one particle touched them both regardless of their being nothing to seemingly connect them. They were created together in the same place, and now something unknown binds them together as a pair at least until their spins are defined. You can subsequently change the spin direction of one of them, and it doesn’t affect the other, so whether this ‘strange glue’ which binds them together somehow disappears once the spin direction is no longer dependent on the other, or whether it forever remains but has no effect, is unknown because we do not know what it is.
It took until 1964 for John Stewart Bell to propose an experiment of a statistical nature which proved that the quantum states of particles could be entangled.
The basis of Bell’s Theorum is difficult to describe concisely without resorting to physics and equations, but there is a good article about it in Quanta Magazine. I cannot explain it any better.
What we know is that there is either some place where the spin of the entangled particles is kept, which both particles can access, or they are able able to communicate over any distance – in both situations instantly. This is contrary to our long-held concpet of spacetime where no communication can take place faster than an electromagnetic wave can travel, in other words the speed of light.
Where Does This Leave Us?
Erwin Schroedinger put forward his thought experiment involving a cat[6], some say to expose how ridiculous quantum entanglement appears to be if applied to everyday objects. Quantum entanglement doesn’t work with larger objects because a large object is made up from smaller particles and doesn’t have a simple set of quantum properties to entangle, all its properties are emergent – they exist due to the behaviour of a collection of particles. Quantum entagnlement has been demonstrated on very small collections of particles, but is unlikely to be possible with anything complex.
To this day nobody has explained how quantum entanglement happens, and it does not fit into many of the models and concepts that form the structure of modern physics. Neither do black holes and neutrinos. This means there is something either missing, or fundamentally wrong with how we understand the universe.
Some now think of everything as being probability waves, and that entanglement is a form of probability wave collapse[7]. This assumes that everything is just an exceedingly complex probability wave that exists everywhere all at once, and the need for anything to travel anywhere is not applicable. In other words everything in the universe is totally and instantly connected. This is a bit like taking us back to saying that everything simply ‘is what it is’, but offers no explanation as to why it is, what these probability waves actually are, or the underlying mechanism that allows them to exist ‘everywhere all at once’. It solves the problem by simplifying the answer to a point where everything is explained by a single mechanism… which remains unexplained. Many people say that this might explain how everything happens – but you’d better be able to prove it because until you do it isn’t science.
What we are discovering in recent times is beginning to put an end to the usefulness of ‘spacetime’ as a model in modern physics. There is a growing feeling that time, distance and speed, and therefore gravity, may be emergent properties which occur only because matter exists, and are not a fundamental property of the universe.
These are other topics entirely, and much still falls under the banner of philosophy rather than science, the difference being whether we can prove these assertions rather than just suspect them.
Featured image: Ashurov sindor, CC BY-SA 4.0 https://creativecommons.org/licenses/by-sa/4.0, via Wikimedia Commons
- [1] Quanta Magazine: https://www.quantamagazine.org/the-electron-is-so-round-that-its-ruling-out-new-particles-20230410/
- [2] JOGOS Public Assets, CC BY-SA 4.0 <https://creativecommons.org/licenses/by-sa/4.0>, via Wikimedia Commons
- [3] Bernard Schaeffer, Public domain, via Wikimedia Commons
- [4] https://cds.cern.ch/record/142461/files/198009299.pdf
- [5] https://arxiv.org/pdf/2507.13582
- [6] New Scientist: https://www.newscientist.com/definition/schrodingers-cat/
- [7] https://pmc.ncbi.nlm.nih.gov/articles/PMC9029371/